
by Nishka Research | Dec 26, 2025 | News
Nishka Research was proud to participate in the 74th Indian Pharmaceutical Congress (IPC) 2025, held in Bengaluru, one of the most significant global gatherings of the pharmaceutical and life sciences community. The event brought together industry leaders, regulators,...

by Nishka Research | Sep 13, 2025 | Knapp Kit
Introduction: In the pharmaceutical world, ophthalmic products (eye drops, intraocular injections, eye ointments, etc.) require exceptional care in manufacturing. The eye is a very sensitive organ, and any impurity in a product intended for ocular use can have serious...
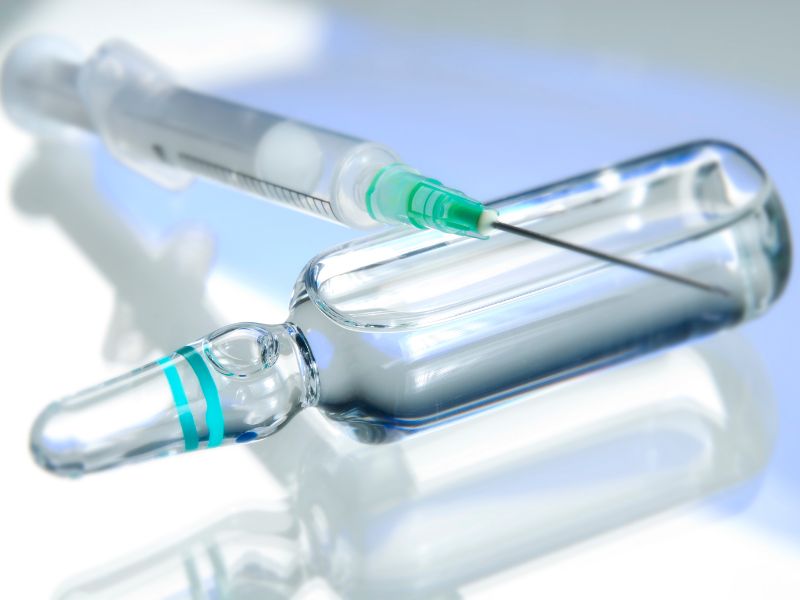
by Nishka Research | Sep 13, 2025 | Knapp Kit
When producing sterile injectable medicines, controlling particulate matter is a critical quality and safety priority. Parenteral product manufacturers worldwide face strict expectations to minimize both visible and invisible (subvisible) particles in their products....

by Nishka Research | Aug 9, 2025 | Knapp Kit
Particulate matter in injectables is one of the most scrutinized quality attributes in sterile drug manufacturing – and for good reason. Visible particles (like lint fibers or glass flakes) floating in an IV bag or syringe can directly endanger patients, causing vein...

by Nishka Research | Aug 2, 2025 | Pharmaceutical
Introduction: In today’s extremely regulated medical device industry, understanding what your device is made of and how those materials behave isn’t just good science – it’s a regulatory and quality imperative. This is where medical device characterization comes into...







