Thinking
Beyond the Box
~ Albert Einstein
Consulting
Helping you realize your product’s potential with scalable, robust solutions.

Testing
Meeting all your testing needs—quality, safety, sustainability, and performance.
Analytical Techniques

We deliver Next-GenResearch Services
Our research processes and solutions are custom based to suit your needs be it in pharmaceutical, biotechnology, manufacturing or any other industry. Our team’s research has demonstrated promising results using cutting edge resources which are unrivalled in the current times.
15 Years of Expertise
Solving Complex Challenges
Advanced Analytical Techniques
AdvancedAnalytical Techniques
Synergism for Collaboration
We collaborate with our clients to produce remarkable developments so that they can achieve their most important goals. NRPLs wealth of established knowledge —and conducting supplementary research when appropriate— we provide evidence based and trustworthy data and meaningful recommendations.

456+
Projects Done

250+
Clients Served
SectorsWe Serve
Uncover the Surface: FE-SEM Case Study of a Cobalt-Chromium Cardiac Stent
Cardiac stents are small, mesh-like tubes implanted in coronary arteries to restore blood flow. Modern stents often use cobalt-chromium
Corneal Lenticule Analysis through the Lens: Integrating Scanning Electron Microscopy and Optical Microscopy Techniques
Introduction At the cutting edge of ophthalmic innovation, Nishka Research presents a pioneering approach to corneal lenticule analysis by
Unlocking Success: Pellet Characterization for Superior Drug Delivery
Introduction: Experience the power of scientific innovation with Nishka Research Pvt Ltd as we unveil a remarkable case study
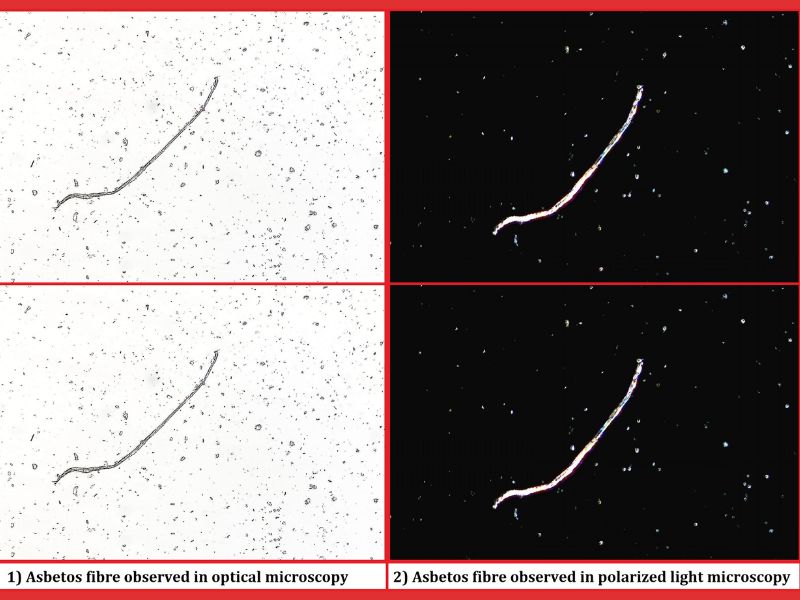
Qualitative Evaluation of Asbestos in Talc: Methods and Techniques
Challenge: A leading cosmetics manufacturer approached Nishka Research for qualitative evaluation of talc samples that were suspected to contain
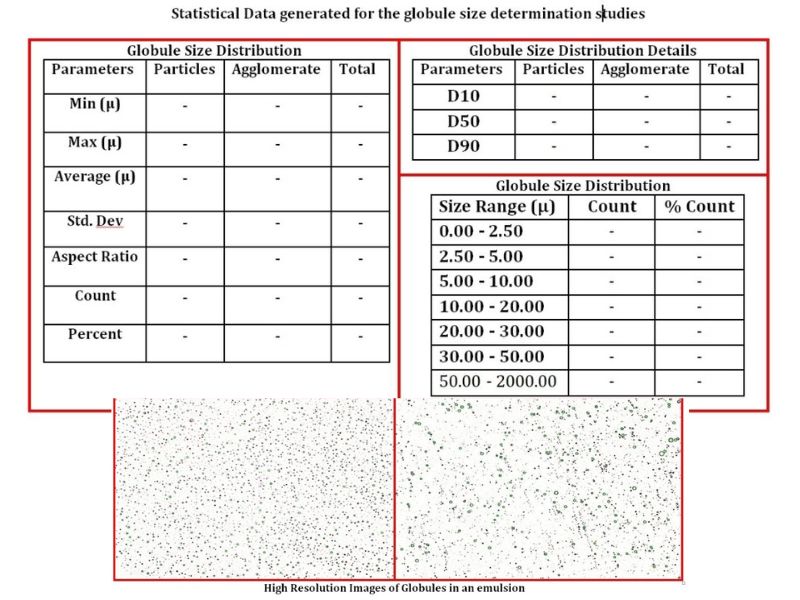
Determination of Globule size in Cream
The Challenge The main challenge for the characterization of Globule size in ophthalmic emulsion (cream) is to evaluate the
Unlock innovation with Nishka Research

OurBlog
Nishka Research @ BioAsia-2026
Great Conversations at BioAsia 2026 – Stall M-19 BioAsia 2026 gave us a strong platform to connect with teams looking to strengthen: People didn’t just walk up to talk about…
Knapp Kits in Inspection Workflows – Manual and Automated Integration Across Pharma Operations
Introduction – Visual Inspection as a GMP Control Point In sterile manufacturing, 100% visual inspection of every container is mandated by GMP standards. Pharmacopeias (USP, Ph. Eur., JP) require that…
Particle, Fiber and their importance in Designing Knapp Kits
Ensuring 100% Visual inspection of sterile products is mandated by almost all regulatory agencies with their respective pharmacopeial and GMP standards. This can be achieved by training and qualifying the…
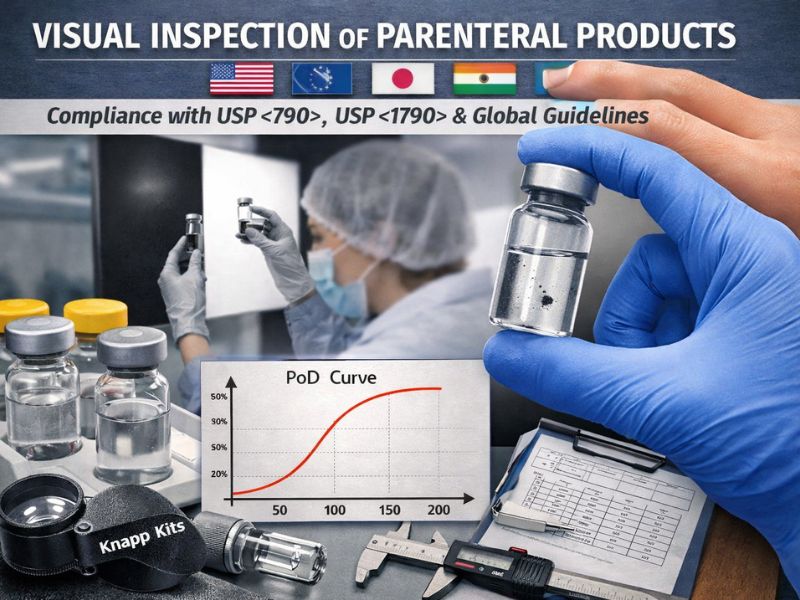
Regulatory Foundations of Knapp Kits: USP <790>, <1790> and Global Compliance
Visual inspection of parenteral products is a critical regulatory requirement worldwide. Standards like USP <790> (“Visible Particulates in Injections”) and USP <1790> (“Visual Inspection of Injections”) codify how manufacturers must…
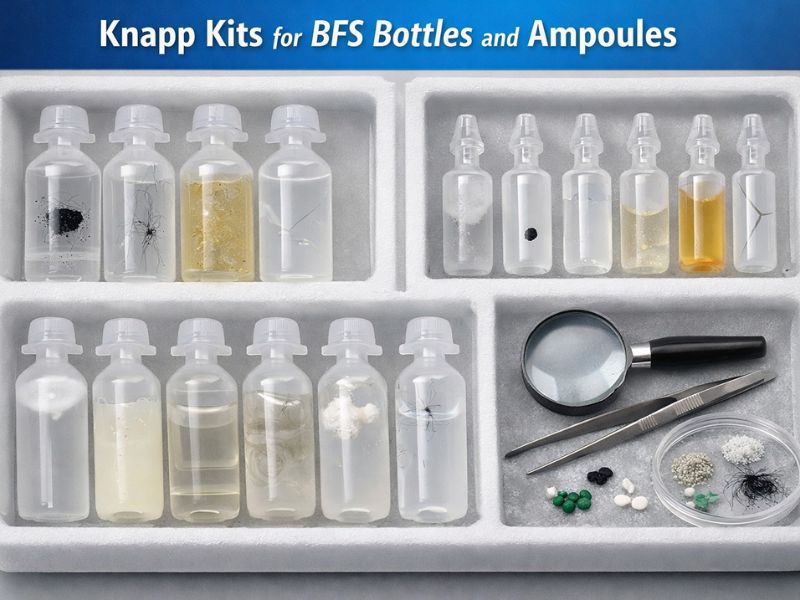
Knapp Kits for BFS (Blow-Fill-Seal) Bottles and Ampoules to ensure visual Inspection Excellence
The most important critical quality control step for injectable and ophthalmic products is Visual inspection. Blow-fill-seal (BFS) is a process, in which the ampoules and bottles are formed, filled and…
Ensuring Ophthalmic Product Quality with Knapp Kits and Visual Inspection
Introduction: Quality and Safety in Ophthalmic Preparations Ophthalmic drug products are applied into eyes, whether eye drops, intraocular injections, or ointments they must meet the highest quality standards to protect…
Nishka Research at the 74th Indian Pharmaceutical Congress 2025, Bengaluru
Nishka Research was proud to participate in the 74th Indian Pharmaceutical Congress (IPC) 2025, held in Bengaluru, one of the most significant global gatherings of the pharmaceutical and life sciences…
Ensuring Ophthalmic Product Safety: The Importance of USP <789> and Manual Microscopic Particle Testing
Introduction: In the pharmaceutical world, ophthalmic products (eye drops, intraocular injections, eye ointments, etc.) require exceptional care in manufacturing. The eye is a very sensitive organ, and any impurity in…
Ensuring Particle-Free Injectables: USP <788> and Global Best Practices
When producing sterile injectable medicines, controlling particulate matter is a critical quality and safety priority. Parenteral product manufacturers worldwide face strict expectations to minimize both visible and invisible (subvisible) particles…
Visible vs. Sub-Visible Particulate Matter: A Critical Quality Concern
Particulate matter in injectables is one of the most scrutinized quality attributes in sterile drug manufacturing – and for good reason. Visible particles (like lint fibers or glass flakes) floating…