Blog
Schedule a MeetingNishka Research @ BioAsia-2026
Great Conversations at BioAsia 2026 – Stall M-19 BioAsia 2026 gave us a strong platform to connect with teams looking to strengthen: Visual inspection performance...
Knapp Kits in Inspection Workflows – Manual and Automated Integration Across Pharma Operations
Introduction - Visual Inspection as a GMP Control Point In sterile manufacturing, 100% visual inspection of every container is mandated by GMP standards. Pharmacopeias...
Particle, Fiber and their importance in Designing Knapp Kits
Ensuring 100% Visual inspection of sterile products is mandated by almost all regulatory agencies with their respective pharmacopeial and GMP standards. This can be...
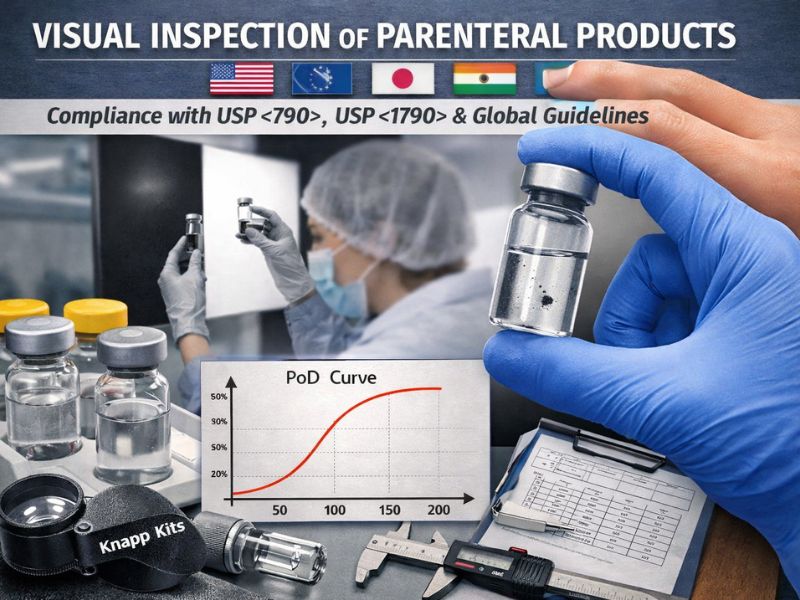
Regulatory Foundations of Knapp Kits: USP <790>, <1790> and Global Compliance
Visual inspection of parenteral products is a critical regulatory requirement worldwide. Standards like USP <790> (“Visible Particulates in Injections”) and USP...
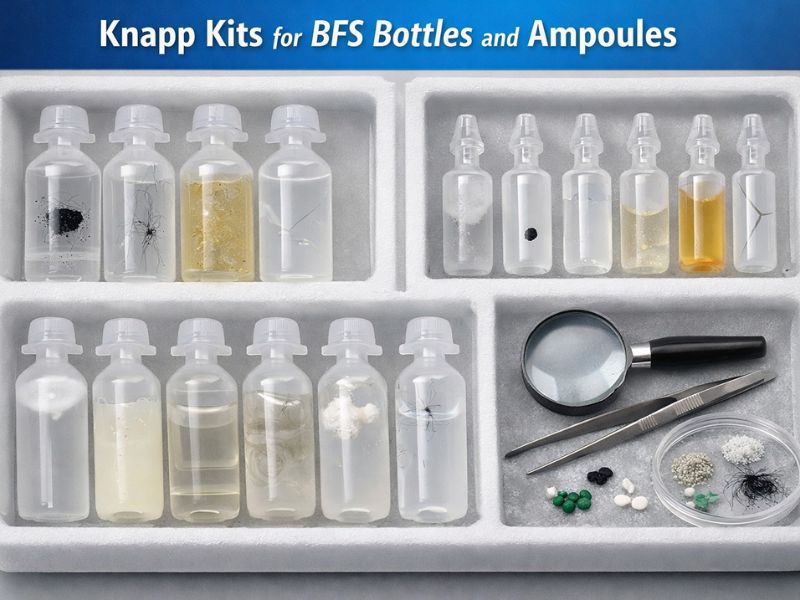
Knapp Kits for BFS (Blow-Fill-Seal) Bottles and Ampoules to ensure visual Inspection Excellence
The most important critical quality control step for injectable and ophthalmic products is Visual inspection. Blow-fill-seal (BFS) is a process, in which the ampoules...
Ensuring Ophthalmic Product Quality with Knapp Kits and Visual Inspection
Introduction: Quality and Safety in Ophthalmic Preparations Ophthalmic drug products are applied into eyes, whether eye drops, intraocular injections, or ointments they...
Nishka Research at the 74th Indian Pharmaceutical Congress 2025, Bengaluru
Nishka Research was proud to participate in the 74th Indian Pharmaceutical Congress (IPC) 2025, held in Bengaluru, one of the most significant global gatherings of the...
Ensuring Ophthalmic Product Safety: The Importance of USP <789> and Manual Microscopic Particle Testing
Introduction: In the pharmaceutical world, ophthalmic products (eye drops, intraocular injections, eye ointments, etc.) require exceptional care in manufacturing. The...
Ensuring Particle-Free Injectables: USP <788> and Global Best Practices
When producing sterile injectable medicines, controlling particulate matter is a critical quality and safety priority. Parenteral product manufacturers worldwide face...
Visible vs. Sub-Visible Particulate Matter: A Critical Quality Concern
Particulate matter in injectables is one of the most scrutinized quality attributes in sterile drug manufacturing – and for good reason. Visible particles (like lint...
Characterization Techniques Every Medical Device Manufacturer Should Know
Introduction: In today’s extremely regulated medical device industry, understanding what your device is made of and how those materials behave isn’t just good science –...
Nishka Research Launches State-of-the-Art FE-SEM with EDAX: Setting New Standards in Analytical Technology
We are proud to announce a landmark enhancement at Nishka Research: the imminent launch of our advanced Field Emission Scanning Electron Microscope (FE-SEM) equipped...