Cardiac stents are small, mesh-like tubes implanted in coronary arteries to restore blood flow. Modern stents often use cobalt-chromium (CoCr) alloys for their platforms, as this material offers high radial strength in a slim profile, allowing thinner struts without sacrificing structural integrity. Thinner struts improve flexibility and vessel coverage, enhancing delivery and potentially clinical outcomes. Ensuring a flawless surface morphology on these stents is critical, because even microscopic imperfections can impact device performance and biocompatibility. In this case study, we explore how Field Emission Scanning Electron Microscopy (FE-SEM) was employed to evaluate the surface of a cobalt-chromium cardiac stent, as part of Nishka Research’s commitment to advanced medical device analysis.
Introduction & Challenge
Our client is a leading global medical device manufacturer, developed a next-generation cobalt-chromium coronary stent and sought independent surface morphology evaluation to verify its quality. The challenge was to confirm that the stent’s laser-cut and electropolished surface was ultra-smooth and free of any micro-scale defects (such as burrs, pits, or cracks) that could jeopardize its performance. A smooth, defect-free stent surface is crucial not only for mechanical reliability but also to reduce the risk of complications like clot formation and inflammation. Conventional optical inspection can miss sub-micron irregularities, so FE-SEM analysis was chosen as the primary technique to scrutinize the stent’s surface at high magnification. This approach aligns with what many medical device organizations ask: How can we ensure our stents have no hidden surface flaws? The solution lies in state-of-the-art microscopy that reveals the stent’s true complexion at the microscopic level.
Our FE-SEM Analytical Approach
We haveimplemented a comprehensive analytical strategy focusing on FE-SEM, complemented by select ancillary techniques for a thorough evaluation:
- FE-SEM Imaging: FE-SEM allowed us to detect any micro-cracks, machining burrs, or other artifacts that could be remnants of the laser cutting or polishing process. FE-SEM’s superior resolution (down to the nanometer scale) ensured that even closely spaced or minute features on the cobalt-chromium stent were clearly resolved, providing a 3D-like view of the surface for in-depth analysis.
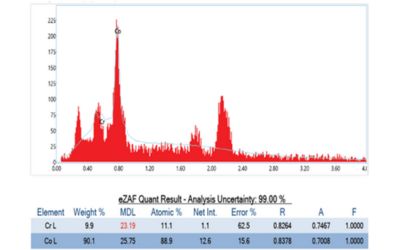
- Elemental Analysis (EDS): An integrated Energy Dispersive X-ray Spectroscopy detector was employed alongside SEM to verify the stent’s material composition. The cobalt-chromium alloy’s elemental makeup was confirmed, and no foreign contaminants or residues were detected on the surface. In the present case, the composition matched the expected CoCr profile, reaffirming material consistency across the stent.
(Note: Prior to FE-SEM, a light optical microscope exam was also performed for a general overview. However, it did not reveal any obvious issues – underscoring the need for FE-SEM’s high magnification to catch what optical methods might overlook.)
Results and Insights
The FE-SEM examination provided a detailed view of the stent’s surface condition. We observed that the cobalt-chromium stent exhibited a uniformly smooth surface finish with well-defined strut edges. Crucially, no micro-defects were found – there were no signs of micro-cracks, jagged burrs, or rough spots on the strut surfaces. The electropolishing step in manufacturing had clearly been effective, yielding a polished surface free of machining debris or sharp artifacts. There was no evidence of pitting or corrosion on the as-manufactured stent.
Through the EDS elemental mapping, we also confirmed homogenous distribution of cobalt and chromium across the stent, with no unusual elemental anomalies. This uniformity suggests consistent alloy properties throughout the device, which is important for predictable mechanical behavior (e.g. uniform expansion during deployment). In summary, the FE-SEM analysis validated that the stent met high quality standards.
Conclusion
This case study highlights how FE-SEM-based surface morphology evaluation can be a game-changer in medical device quality assurance. By leveraging advanced microscopy, Nishka Research was able to peel back the layers of the cobalt-chromium of cardiac stent’s surface, revealing a reassuring picture of structural integrity and precision engineering. The stent’s ultra-smooth, defect-free surface (verified at nanoscopic resolution) is expected to support its safety and efficacy in patients – minimizing risks of thrombosis and fostering better endothelial healing post-implantation.
From an industry perspective, using FE-SEM for stent analysis addresses common questions posed by medical device organizations: “Are our manufacturing processes yielding the desired surface quality? Are there any hidden flaws that could cause failures?” Our findings in this real-world scenario provided a clear answer, enabling the manufacturer to proceed with confidence in their product’s quality.



0 Comments