
by Navya Sri | Feb 24, 2026 | Knapp Kit, Pharmaceutical
Great Conversations at BioAsia 2026 – Stall M-19 BioAsia 2026 gave us a strong platform to connect with teams looking to strengthen: Visual inspection performance Foreign particle investigations API particle size distribution studies & morphology understanding...

by Navya Sri | Jan 17, 2026 | Knapp Kit
Introduction – Visual Inspection as a GMP Control Point In sterile manufacturing, 100% visual inspection of every container is mandated by GMP standards. Pharmacopeias (USP, Ph. Eur., JP) require that final products be “essentially free” of visible defects...

by Navya Sri | Jan 12, 2026 | Knapp Kit
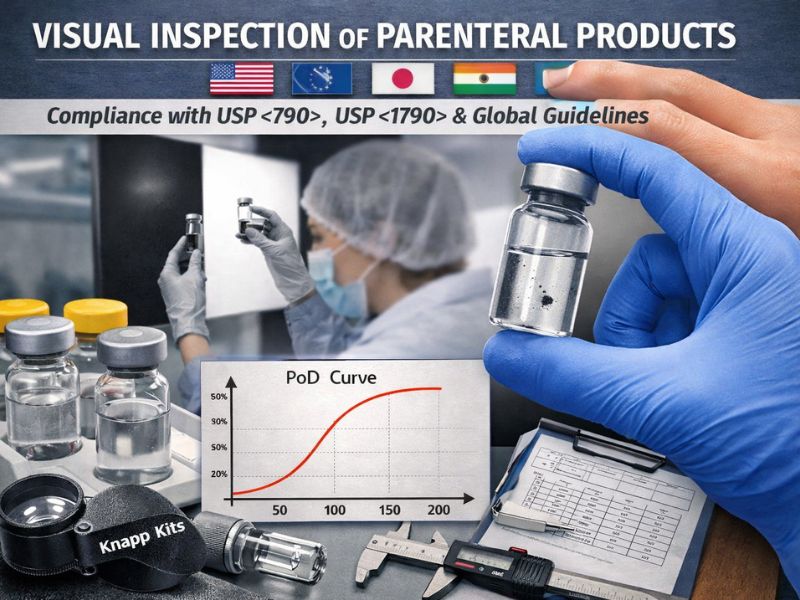
Ensuring 100% Visual inspection of sterile products is mandated by almost all regulatory agencies with their respective pharmacopeial and GMP standards. This can be achieved by training and qualifying the visual inspectors using Knapp Kits or challenge kits which are...
by Navya Sri | Jan 12, 2026 | Knapp Kit
Visual inspection of parenteral products is a critical regulatory requirement worldwide. Standards like USP <790> (“Visible Particulates in Injections”) and USP <1790> (“Visual Inspection of Injections”) codify how manufacturers must inspect each container...
by Navya Sri | Jan 3, 2026 | Knapp Kit
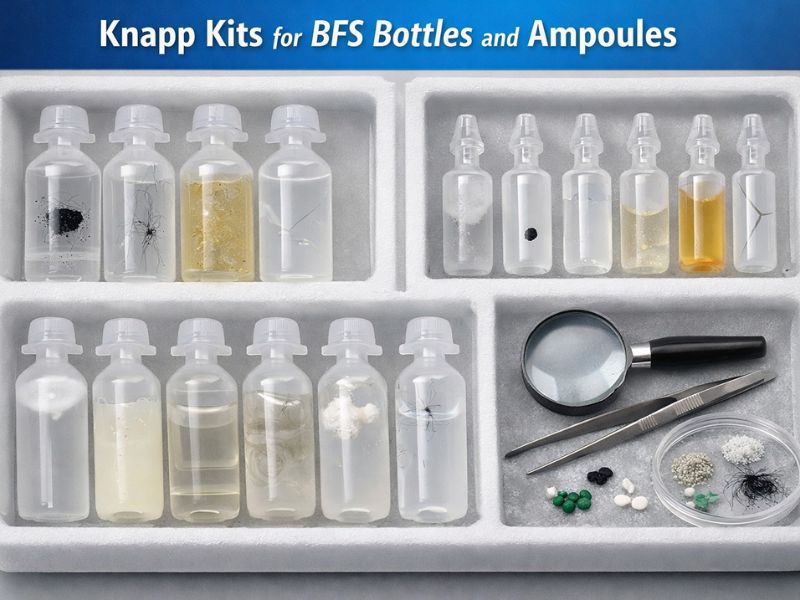
The most important critical quality control step for injectable and ophthalmic products is Visual inspection. Blow-fill-seal (BFS) is a process, in which the ampoules and bottles are formed, filled and sealed in single automated device, which ensures each unit is...

by Navya Sri | Dec 31, 2025 | Knapp Kit
Introduction: Quality and Safety in Ophthalmic Preparations Ophthalmic drug products are applied into eyes, whether eye drops, intraocular injections, or ointments they must meet the highest quality standards to protect patients’ vision and health. Even tiny...








