The most important critical quality control step for injectable and ophthalmic products is Visual inspection. Blow-fill-seal (BFS) is a process, in which the ampoules and bottles are formed, filled and sealed in single automated device, which ensures each unit is essentially free of visible defects and particles. Global regulators require 100% inspection of parenteral containers and Knapp kits have emerged as indispensable tools to qualify both MVI (manual visual inspectors) and AVI (automated visual inspection systems) in meeting these standards. This comprehensive guide explores BFS technology applications (unit-dose ampoules, multi-dose bottles and ophthalmic droppers), the unique inspection challenges in BFS (especially for biologics), the role of Knapp test kits in training and validation, and the expectations of regulatory agencies like the USFDA, EMA, PMDA, WHO and CDSCO. By understanding these aspects, pharmaceutical and biotech manufacturers can optimize their BFS inspection programs to ensure patient safety and regulatory compliance.
Understanding BFS Technology and Its Applications
Blow-Fill-Seal (BFS) is an advanced aseptic manufacturing technology where a plastic is blown to form a container, immediately filled and then sealed in one continuous operation. This seamless process minimizes human intervention and contamination risk. BFS has become popular for a range of sterile products, including small-volume injectables and ophthalmics, due to its efficiency and safety benefits. Key BFS packaging formats include –
- Unit-Dose BFS Ampoules – Small (often 0.5 mL to 10 mL) single-use ampoules commonly used for eye drops, respiratory solutions (nebulizer vials), vaccines, and flush solutions. These ampoules are typically linked in strips and opened by twisting off a tab. Their one-time-use design eliminates preservatives, making them ideal for sensitive formulations (e.g. preservative-free ophthalmics).
- Multi-Dose BFS Bottles – Larger volume BFS containers (e.g. 30 mL, 100 mL or more) that serve as multi-dose bottles or infusion containers. Examples include BFS bottles for IV solutions or irrigation fluids, and ophthalmic multi-dose droppers that incorporate special valves to remain sterile across uses. BFS bottles can be designed with integrated caps or ports, allowing repeated access when needed.
- BFS Ophthalmic Containers – BFS is widely used for ophthalmic products, both in unit-dose form (single-use droppers) and multi-dose form. The BFS process ensures sterility of eye drop solutions without requiring a separate sterilization step. Ophthalmic BFS ampoules are often packaged in strips of multiple droppers, while multi-dose BFS eye drop bottles may include built-in applicators. These formats must be carefully inspected for clarity and particulate absence to protect delicate ocular tissues.
BFS offers advantages like shatterproof containers (no glass breakage), reduced particulate contamination (no glass or rubber shedding), and high sterility assurance due to the closed, aseptic formation process. Catalent’s Advasept® BFS technology, for instance, has been used for biologic drugs (large proteins) and demonstrated reduced risk of contamination and no glass delamination. Moreover, BFS allows custom container designs (e.g. reduced headspace) which can lessen product agitation and thereby improve stability for sensitive biologics. In short, BFS ampoules and bottles have diverse applications across injectables, biologics, and ophthalmics, but with these benefits come unique challenges in ensuring quality through visual inspection.
Visual Inspection Challenges in BFS Packaging
Inspecting BFS containers for defects and particles is inherently challenging due to the nature of the materials and products involved. Unlike clear glass vials, BFS ampoules and bottles are made of semi-transparent plastic (typically polypropylene or polyethylene). This reduced clarity means that detecting tiny particles is more difficult – in fact, the visible particle size threshold in BFS containers is larger compared to glass vials. In practice, an inspector or camera might only catch larger particulates in a translucent BFS ampoule that would have been visible at smaller sizes in a clear glass vial. This limitation must be accounted for in inspection programs (e.g. by defining a higher size threshold for “visible” particles in BFS).
At the same time, the entry of extrinsic particles is reduced a lot as the container is formed, filled and sealed in a sterile system within seconds, there is minimal exposure to environmental contaminants.
Literature suggests that there is “a lower risk” of particulate contamination when compared to glass container, as glass containers have few additional steps like container washing, sterilization and handling.
So the choice would be BFS containers which have fewer chances of particles entry, however BFS packaging pose another challenge that is identification of particles in BFS
Few other inspection challenges of BFS packaging include –
- Container Geometry – BFS ampoules often have connected strips of units, weld seams or tabs. These features may obscure view and may create false shadows during inspection. Inspectors must manipulate BFS units (rotate, squeeze, etc.) to view all sides. Automated systems need multiple camera angles or imaging techniques to achieve full coverage.
- Solution Characteristics – Many BFS-packaged products are not simple clear solutions. Foaming solutions, suspensions, or emulsions increase inspection difficulty, as bubbles or opacity can mask particles. For example, an inhalation suspension in a BFS vial may leave residue on the walls, making it hard to distinguish a contaminant. Inspection sensitivity is inherently lower for non-clear solutions. This must be considered when qualifying inspectors or machines – e.g. setting realistic defect detection thresholds for such products.
- Common Defect Types – BFS containers have their own spectrum of defects to watch for
- Particulate matter – Any visible particle is a potential hazard. This includes extrinsic particles (e.g. fiber, metal, plastic shavings that may enter during processing) and intrinsic/inherent particles (e.g. undissolved drug, precipitates, or protein aggregates from the formulation). Through risk analysis, manufacturers determine likely particle sources in BFS (filler mandrel metal, gasket debris, plastic flash from molding, etc.) and design detection for those. Notably, even protein aggregates (inherent to biologic products) are treated as unacceptable visible particulates by regulators.
- Cosmetic and container defects – BFS ampoules can exhibit cosmetic flaws like scuffs, scratches, or incomplete formation (e.g. a malformed neck or cap). Cracks or pinholes in the plastic, while rare due to the integral molding, can occur and would compromise sterility. Seal defects (improper sealing of the ampoule tip or bottle port) are critical to catch. Additionally, BFS containers may show fill volume deviations if the filling phase had variability, so under-filled or overfilled units must be identified.
- Closure issues – Some BFS bottles include an inserted cap or tip after forming. Misapplied caps, missing or misformed twist-off tabs, or improperly engaged valves (for ophthalmic droppers) are defects to detect. These could lead to leaks or dosing problems.
- Foreign discoloration or fiber – Occasionally, a foreign fiber or a discoloration (e.g. from overheated plastic) might be present, which inspectors need to differentiate from product-related artifacts.
- False Rejection Trend – Literature suggests that most of the false reject trends are observed on BFS ampoules, for instance, a tiny air bubble or a bit of lint on the container surface might be mistaken for a particle inside the container. Also, BFS often generates air bubbles in the liquid during sealing, these bubbles are not harmful but they could be misinterpreted as particulates by human inspectors or automated cameras. This issue is will escalate to Ensure a robust defect library and inspector training (so they don’t erroneously reject bubbles as particles and false rejections can be reduced) is important. AVI algorithms also must be tarined to differentiate moving particles vs. static bubbles.
Despite these challenges, modern inspection solutions can achieve high reliability even for BFS. Rommelag, the inventor of BFS technology, developed a Particle Inspection Machine (PIM) that uses innovation to overcome the lack of container transparency. Instead of spinning the container (as is done with glass vials), their system vibrates BFS ampoules on a block to agitate any particulates in the liquid. Cameras then capture images from the bottom and top to detect particles that either sink or float. This allows 100% inline inspection at full production speeds, with sensitivity surpassing human inspectors. Figure 1 illustrates an automated BFS inspection system – multiple cameras examine each container after a vibration step, identifying moving particulates as defects.

In summary, BFS containers demand careful inspection strategies that acknowledge material and product differences. With appropriate background conditions, defect libraries, and possibly advanced tools (like vibration and imaging algorithms), manufacturers can reliably detect critical defects in BFS ampoules and bottles, ensuring patient safety.
Manual vs. Automated Visual Inspection in the BFS Context
Historically, manual visual inspection (MVI) has been the mainstay for BFS products. Trained human inspectors examine each ampoule or bottle, often holding it in front of a bright white panel for ~5 seconds, then in front of a black panel, looking for any floating or dark particulate. The process is inherently subjective and fatiguing – operators require concentration and must be cycled out frequently to avoid misses due to tired eyes. Industry practice sets a performance benchmark using challenge test sets – for example, inspectors should detect certain standard particles at least 70% of the time to be considered qualified. These test sets (like Knapp kits) contain known defective samples and help ensure that manual inspection isn’t merely cursory but has a quantifiable detection capability.
However, manual inspection has limitations, especially for BFS. The plastic container variability (some may be hazier than others), small particulates that approach the threshold of human vision, and the need to inspect large volumes at speed (a BFS machine can produce thousands of ampoules per hour) all make human inspection less efficient. A human might inspect 100–200 units per minute at best, and their accuracy can drop with fatigue or monotony. Regulators recognize these challenges – USP <1790> suggests that inspectors have periodic retraining and vision tests, limited shifts (typically 20-minute sessions with breaks), and that any manual inspection process is inherently “probabilistic” – meaning some defects can be missed.
Automated Visual Inspection (AVI) systems for BFS have been a game-changer. Modern AVI machines use high-resolution cameras, lighting systems, and sometimes robotics or special handling to inspect each container in a consistent manner. For BFS ampoules, which often come in strips, machines either inspect them in card form or after separating the ampoules. Some key aspects of automated BFS inspection –
- Static vs Dynamic Inspection – Some systems use a static approach – holding the BFS container still and using image processing (frame subtraction, particle movement detection) to find anomalies. This is often simpler mechanically and suitable for BFS, as demonstrated by Vitronic’s BFS inspection that uses static imaging with minimal moving parts. Others use a dynamic approach like the vibration method mentioned earlier or spinning larger BFS bottles to swirl the liquid. Both aim to differentiate actual particles (which move under agitation) from stationary background.
- Multiple Camera Stations – To fully cover a BFS container, AVI machines have multiple stations – e.g. one camera checks the tip/cap area, another scans the container body and seams, another looks at the bottom for sediments. They also often integrate fill-level checks, verifying the correct volume in each container by analyzing the meniscus position or using sensors. Any cosmetic defects (like severe scratches or discoloration of plastic) can be detected by surface inspection cameras as well.
- Reject Mechanisms – When a defect is identified in an ampoule on a strip, the system will typically mark or segregate that strip. Downstream equipment then removes the entire strip or that particular unit if feasible. High-speed machines coordinate this so that only failing units are rejected, minimizing waste.
- Data and Analytics – Automated systems provide quantitative data – counts of each defect type, trending of reject rates, and even images of each reject. This data can be invaluable for continuous improvement. For example, an increasing trend in “fiber” defects might indicate a filter shedding issue, prompting maintenance before it becomes critical. The data also supports compliance by documenting that inspection is effective and within false reject limits.
Both MVI and AVI systems are used in BFS packaging, but automation is increasingly favored for its efficiency and reliability. Whichever method is used, regular qualification is required – and that’s where Knapp kits come in, ensuring that inspectors and machines are rigorously tested and qualified to catch defects consistently.
Knapp Kits for BFS Ampoules and Bottles – What Are They and Why Use Them?
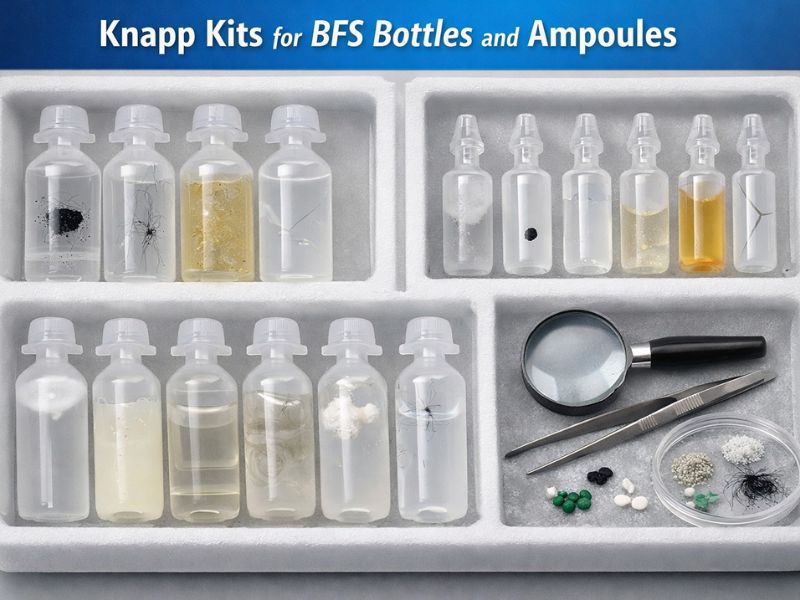
As introduced earlier, Knapp kits (also called visual inspection challenge kits) are standardized sets of containers deliberately prepared with known defects. They are used to train, qualify, and challenge both human inspectors and automated inspection machines. In pharmaceutical QC, “Knapp kits are essential tools… enabling manufacturers to ensure packaging integrity, patient safety, and compliance with international regulations”. In practical terms, a Knapp kit for BFS ampoules/bottles might consist of dozens of BFS containers – some that are “good” (defect-free) and some that contain specific defects of calibrated size/type (e.g. a 150 µm fiber in solution, a piece of metal shard, an under-filled unit, a container with a micro-leak, etc.). The composition of the kit and the test protocol are carefully designed so that one can assess an inspector’s or machine’s detection capability statistically.
How Knapp Kits are Used –
- Inspector Training and Qualification – New inspectors learn using Knapp kits by examining these samples and trying to find all the defects. To become qualified, an inspector typically must achieve a certain detection score (e.g. detect ≥80% of the defects, with ≤5% false positives) in multiple trials. USP <1790> recommends a life-cycle approach where inspectors are qualified initially and then re-qualified (e.g. annually) using challenge kits. The kits help standardize training – every inspector faces the same level of challenge, reducing subjectivity. If an inspector fails to detect enough defects (or incorrectly rejects too many good units), they receive further training or may be disqualified from inspection duties until issues are resolved.
- Automated Machine Validation (Knapp Test for AVI) – To validate an automatic visual inspection system (AVI) for BFS, the machine is tested with a Knapp kit as well. A side-by-side study is done where a set of samples is inspected by qualified humans and by the machine. If the machine catches as many or more defects than humans (and stays within allowable false reject rates), it “passes” and can be used in production. This is essentially the Knapp Test protocol, which is even referenced in regulations. For instance, the EU Annex 1 indicates using “representative defects” to challenge equipment performance at startup and regular intervals. The Knapp test verifies the AVI is not missing anything the human eye would catch, thereby justifying that 100% inspection can be reliably done by machine.
- Calibration of Detection Thresholds – Knapp kits often include defects of varying sizes to pinpoint the detection threshold – the smallest or faintest defect that can be consistently detected. For example, a BFS kit might have particles of 100 µm, 150 µm, 200 µm, etc. If inspectors reliably catch the 200 µm but only 50% of the 150 µm, one might set ~200 µm as the effective threshold in that scenario. Such information is critical for risk assessments (knowing what size of particle might escape detection) and is used to improve processes or enhance inspection if needed. The FDA has noted that “threshold studies should be conducted to determine the size of visible particulates that can be reproducibly detected by trained personnel”, reinforcing this approach.
- Ongoing Performance Monitoring – Beyond initial qualification, some firms employ challenge kits periodically (e.g. daily or weekly) on inspection lines to ensure nothing has drifted. For example, running a few defect samples through an automated BFS inspection machine at the start of each batch can confirm the system is still detecting properly (a form of continuing calibration). Any failure to detect a test defect would trigger an investigation or recalibration. This practice aligns with GMP expectations of continuous assurance – not just qualifying once and assuming perpetual performance.
Knapp Kits for BFS vs. Other Formats – A Knapp kit must use containers and defects that closely resemble the actual product being inspected. There are only few suppliers for BFS Knapp kits especially reputed Knapp kit manufacturers like Nishka Research Pvt. Ltd., offer customized Knapp kits for BFS. Nishka research website claims that their customized Knapp kits resemble the actual BFS production line. Using a conventional Knapp kit (Like glass vial kit to qualify a BFS line) will not address the issue, as the optical properties of BFS differ. As per the of USP <1790> and EU Annex 1) which emphasizes using appropriate sample sets during qualification, the samples should be “as similar as possible in shape, size, color, and product to those inspected routinely”. BFS kits meet this need by providing defect samples in actual BFS ampoules or bottles made of the same plastic and typically produced on BFS equipment.
- Contents of a Knapp Kit – While specifics vary, a comprehensive BFS Knapp kit could include. Particle Defects – Vials with inserted particles (e.g. a stainless-steel particle of known size, a fiber, a rubber fragment if relevant, a speck of plastic, etc.) suspended in the liquid. These test the detection of foreign particulates, a major concern for injectables.
- Cosmetic Defects – Containers with visual flaws like a crack, a bubble in the wall, a large scratch, or discoloration. These help ensure inspectors also pay attention to container integrity and appearance, not just particles.
- Fill Volume Defects – Some units filled intentionally high or low to test fill-level detection systems or the acuity of inspectors in spotting volume deviations.
- Seal/Closure Defects – For instance, a bottle with a pinhole leak or an ampoule with an incompletely sealed tip. These can be hard to spot by eye (often manifesting as a subtle liquid weep or a tiny void at the seal), so including them challenges the inspection process’s ability to catch leaks. Often these are also checked via other methods like leak testing, but visual inspection is the primary screen for seal integrity immediately after filling in many BFS operations.
- Good Units – A majority of the kit may be comprised of “good” containers with no defects. These establish the background false reject rate; an inspector or machine should not erroneously flag too many good units. A false reject rate (FRR) of ≤5% is a typical benchmark in industry qualification, meaning at most 5 out of 100 perfect units might be wrongly rejected.
- Each defect in the kit comes with documentation – typically a certificate or data sheet describing the defect (size, type) and often microscopic images for reference. Kits are usually traceable to standards. They also have an expiration of 2 years, since over time the defects could change (particles might dissolve or relocate, etc.). Proper handling (e.g. gentle mixing before use to ensure particles aren’t stuck to walls) and storage conditions are defined to keep the kit valid.
Global Regulatory Expectations (USFDA, EMA, PMDA, WHO and CDSCO) for BFS Inspection
Visual inspection requirements for parenteral drugs are stringent worldwide, and BFS products are no exception. While not every agency issue BFS specific guidance, all major regulators mandate that injectable product units be 100% visually inspected and that processes be validated. Below is an overview through a regulatory lens, focusing on the United States and also covering Europe, Japan, WHO guidelines, and India.
- United States (FDA & USP) – The FDA’s current Good Manufacturing Practices (cGMP) and USP standards form the basis of U.S. expectations. USP <790> states that “all parenteral products should be essentially free from any visible particles.” This is echoed across pharmacopeias as a fundamental requirement – no vial or ampoule should have observable contaminants. This can be achieved by 100% inspection of each filled container before distribution. The FDA enforces this through 21 CFR parts 210/211 (though not explicitly spelled out, it’s implicit in requiring freedom from particulates and in acceptance testing of each batch). As per the guidance released in 2021 “Inspection of Injectable Products for Visible Particulates,” which importance to holistic, risk-based approach to address the particulate control from development through production. Notably, FDA clarifies that merely meeting USP compendial tests (like sampling a few units for visible particles) “is not generally sufficient” to meet GMP, manufacturers need a robust program including thorough inspection, investigations of root causes and preventive measures. The guidance also encourages use of scientifically sound methods for qualification (implicitly supporting things like Knapp tests for setting detection thresholds and assessing inspector performance). FDA expects any AVI systems must be qualified and shown to perform as well as an experienced human inspector, which aligns with industry practice of the Knapp test. During inspections, the FDA will look for evidence of inspector training, regular vision acuity tests and defect libraries or test sets used for qualification. A common FDA observation in the past has been failure to adequately detect and remove particulate contaminants, hence companies place great emphasis now on this area, particularly for sensitive biologic injectables.
- Europe (EMA and EU GMP) – The European Medicines Agency, via the EU GMP guidelines (especially Annex 1 – Manufacture of Sterile Products), has very explicit requirements on visual inspection. The latest revision (effective 2023) states – “All filled containers of parenteral products should be inspected individually for extraneous contamination or other defects.”. It requires that manufacturers establish a defect classification (critical, major, minor) and maintain a defect library for training purposes. Annex 1 also mandates annual re-qualification of operators under worst-case conditions (including end-of-shift fatigue, line speeds, etc.). For automated systems, as mentioned, Annex 1 8.32 requires validation proving the machine is “equal to or better than” manual inspection, and ongoing challenge tests with samples. EU inspectors (and national authorities like Germany’s BfArM, France’s ANSM, etc.) will expect to see evidence of these practices. In fact, a review of EU inspection findings showed many deviations related to inadequate staff qualification and lack of defect kits. This highlights that EU regulators pay close attention to whether firms use something like Knapp kits and have clear criteria for inspector performance (e.g. what % of defective units must be detected during qualification). The Ph. Eur. (European Pharmacopoeia) also has general chapter 2.9.20 on visual particulates, aligning with USP <790> in that injections should be practically free of visible particles – reinforcing that 100% inspection is expected to ensure this outcome. In summary, Europe’s regulatory lens demands a highly controlled visual inspection process with training, defect libraries, and validation all documented – with BFS containers being treated just like any other injectable in this regard.
- Japan (PMDA & JP) – Japan’s PMDA adheres to standards comparable to the U.S. and EU. The Japanese Pharmacopoeia (JP) has provisions similar to USP <790> (since Japan is part of ICH and harmonizes many quality requirements). Injectable products in Japan must pass tests for visible foreign matter, and 100% inspection is performed. The PMDA inspectors likely reference both JP standards and PIC/S GMP guidelines (Japan is a PIC/S member, which means alignment with EU GMP largely). Thus, it’s expected that Japanese manufacturers also utilize inspector qualification and Knapp-style testing. Notably, many Japanese firms were pioneers in automated inspection technology, so PMDA is well aware of the Knapp test principles. While specific Japanese guidance on BFS per se is not separate, any BFS product (like a BFS-packaged ophthalmic solution) must meet the same criteria of no visible contaminants. PMDA would also expect control of particulates and container defects; for biologics, the Japanese regulators are particularly cautious about protein aggregates as visible particles due to their potential immunogenicity. In short, PMDA’s expectations align with FDA/EMA – ensure an robust visual inspection program, validated and with trained staff.
- World Health Organization (WHO) – The WHO GMP guidance for sterile pharmaceuticals (Annex 6 of WHO Technical Report Series) explicitly mirrors others – “Filled containers of parenteral products should be inspected individually for extraneous contamination or other defects.” and if done visually, it must be under proper lighting and background. It also mentions that if other methods are used (automated), they should be validated and regularly checked. The WHO guidance emphasizes inspector conditions – regular eye tests and frequent breaks are recommended to maintain inspection effectiveness. Many countries’ national regulations (especially in the developing world) are built on WHO GMP guidelines. Therefore, a manufacturer following FDA/EU practices for BFS inspection would inherently satisfy WHO expectations. WHO does not specifically call out “Knapp kits” by name, but the concept of having prepared samples for training and qualification is consistent with GMP. For instance, WHO inspectors would expect to see that operators know what defects to look for and have been tested on their ability to detect them – which is exactly what Knapp kits facilitate.
- India (CDSCO) and Other Agencies – India’s CDSCO (Central Drugs Standard Control Organization) follows a blend of Indian GMP (largely aligned to WHO GMP) and has adopted parts of PIC/S. India’s pharmaceutical industry, being a major global supplier, often voluntarily complies with USP/EP standards as well. The Indian Pharmacopoeia (IP) contains tests for particulate matter and likely the visible particulate requirement similar to USP <790>. Thus, an injectable made in India must be essentially free of visible particles, requiring 100% inspection. CDSCO inspectors, as well as those from other countries like Canada (Health Canada GUI-0119), Australia (TGA), etc., all converge on the same principles – every unit inspected, defects trended, and inspection processes validated. Many of these agencies explicitly list USP <1790> and EU Annex 1 as references for best practices. For example, a supplier of inspection kits notes their kits ensure compliance with “US FDA, EU GMP, JP, MHRA, TGA etc.”—underscoring that the global regulatory framework on this topic is harmonized. Manufacturers exporting to multiple markets must therefore meet the highest common standard, which practically means adhering to the strictest elements of FDA and EU requirements.
It’s worth highlighting that for biologic products (which regulators classify often as high-risk), the scrutiny on visible particles is even greater. Agencies like FDA and EMA require that any inherent protein aggregates that become visible are investigated and minimized through formulation or process tweaks. A BFS container holding a biologic injectable, for instance, might need special inspection considerations (perhaps an additional manual check under magnification for any protein filaments, etc.). Regulators might also expect more frequent requalification of inspectors for these products given the difficulty in distinguishing protein particles from other matter.
In conclusion, across FDA, EMA, PMDA, WHO, and CDSCO, the message is uniform – 100% visual inspection is mandatory, and the process must be controlled, qualified, and documented. Knapp kits and rigorous training programs are a practical way industry meets these regulatory expectations. BFS ampoules and bottles are held to the same standard of “essentially free of visible particulates” as any other injectable container – thus manufacturers must implement robust inspection regimes and demonstrate their effectiveness through the kinds of measures discussed above. Failing to do so can result in regulatory actions (warning letters, recalls) as history has shown. Conversely, a company that uses Knapp kits to qualify inspectors and machines, maintains a defect library, and keeps detailed inspection records will find themselves well-prepared for audits and, most importantly, will consistently deliver safe, quality products.
BFS Inspection Challenges Unique to Biologics
One special area worth delving into is the application of BFS technology to biologic products (e.g. protein therapeutics, vaccines, cell therapies) and the unique inspection challenges they pose. Traditionally, high-value biologics like monoclonal antibodies have been filled in glass vials or pre-filled syringes. However, innovations like BFS sterile vials (e.g. Catalent’s Advasept®) are bringing BFS into the biologics space. When biologics meet BFS, the following considerations become critical –
- Protein Aggregates as Particulates – Biologics are large, complex molecules prone to aggregation (clumping together) under stress (heat, agitation, etc.). These aggregates can become visible particles in the solution. Unlike a speck of dust, these are “inherent” defects (coming from the product itself rather than external contamination), but regulators still require their removal because visible aggregates can harm patients (e.g. by triggering immune reactions). Detecting protein aggregates can be tricky – they might be translucent or form only under certain conditions (e.g. after shipping or temperature changes). In a BFS container, if the plastic is less clear, a faint flocculent aggregate could be overlooked. Inspection systems must be calibrated to detect even these inherently generated particles. This might mean inspecting under varied lighting or even using polarizing filters to spot protein strands. Additionally, aggregation visibility could change with storage, a solution that was clear at filling might develop flakes over time. Quality control might need to inspect products at end of shelf life as well, or use adjunct methods like sub-visible particle testing, but the initial visual inspection remains crucial as a last line of defense before release.
- Temperature Sensitivity – Many biologics are heat-sensitive. BFS involves molding plastic at high temperatures (often >150°C) and immediately filling the product into the hot container. This raised concern that biologic formulations might be exposed to higher temperatures briefly during the BFS process. Indeed, one study noted “the molded plastic reaches elevated temperatures during filling, which may compromise the stability of biologic formulations”. For instance, if a protein gets heated near 40°C or higher, it might start to unfold or aggregate. To mitigate this, BFS providers have optimized processes (e.g. cooling molds, shortening exposure time) – Catalent reported getting solution temperatures down to ~40°C for a 0.5 mL fill, with temperature dropping quickly after sealing. Still, manufacturers must verify that the BFS process doesn’t induce unacceptable changes in the biologic. Visible inspection plays a role here – an increase in aggregates or precipitate in BFS vials versus glass vials could be a red flag that the process is affecting the product. Inspectors might notice slight opalescence (a haze) indicating early aggregation. Such observations would prompt further analytical tests. Thus, inspection personnel for biologics in BFS should be trained to be extra vigilant for signs of product instability (not just foreign contaminants).
- Extractables and Leachables – Plastic containers can release chemical substances (additives, monomers, etc.) into the product, especially under heat or over time. Biologics can be particularly sensitive to such leachates, for example, certain leachables might cause protein aggregation or denaturation. While extractables/leachables studies are primarily chemical tests, they intersect with visual inspection because a severe leachable issue could manifest as visible haze or particles (for instance, if a leached chemical reacts and precipitates). In summary, while not a direct part of routine inspection, the potential for extractables or oxidative particles means inspection protocols for biologics need to account for any unusual defect types (e.g. a fine precipitate that might coat the walls).
- Special Defect Kits for Biologics – If a manufacturer knows that their biologic can form, say, crystalline precipitates under stress, they might include such a scenario in their defect library for training. BFS even offers some benefits to biologics – no glass means no risk of glass flakes or breakage, and reduced headspace can minimize agitation-related aggregation. Nevertheless, it requires a careful marriage of formulation science and inspection rigor. Quality systems should integrate the knowledge of formulation behavior into their inspection training and defect criteria. Any unusual defect observed in a biologic BFS batch (say a cloudy unit) should trigger immediate lab investigation (particle identification, etc.) rather than being dismissed, because it may indicate a formulation issue rather than external contamination.
Conclusion
BFS technology is increasingly popular in pharmaceutical manufacturing, providing sterile ampoules and bottles efficiently and safely. Visual inspection, whether done by skilled manual inspection team or by advanced AVI system, remains the cornerstone of verifying BFS product quality.
In this blogpost, we explored how Knapp kits serve as a powerful enabler for maintaining high inspection standards. By offering a calibrated challenge, these kits help train inspectors, validate automated systems and ultimately prove to regulators that a manufacturer can reliably detect any unit that doesn’t meet specifications. For BFS ampoules and bottles, including those used in unit-dose vaccines, multi-dose eye drops and cutting-edge biologic formulations, Knapp kits tailored to their unique characteristics ensure that the subtle defects not apparent in plastic or under certain solutions are not missed. We’ve seen that global regulatory agencies (US FDA, EU EMA, Japan PMDA, WHO, India CDSCO, and others) all converge on the expectation of thorough visual inspection with documented proof of its effectiveness. Compliance is not optional; it’s a fundamental GMP requirement to inspect each and every container.
Implementing a successful BFS inspection program is a multifaceted effort. It involves engineering (having the right lighting, background, and if automated, the right camera setup), human resources (training and qualifying staff with robust methods), and quality culture (treating each reject or unusual observation as valuable information to improve the process). Knapp test kits and defect libraries transform what could be a subjective process into a quantifiable, continuously improvable system. They help answer questions like – How small a particle can we catch in this 5 mL BFS ampoule? Are we as good this year as we were last year at finding defects? Is our new automated inspector better than the manual method it replaced? The answers to these have direct patient safety impact, and regulators will ask them if you don’t.
Furthermore, as pharmaceuticals evolve – with more sensitive biologics and more reliance on innovative packaging like BFS – the inspection techniques and qualification approaches must evolve too. Challenges such as detecting protein aggregates in BFS containers highlight the need for cross-disciplinary awareness -inspectors should coordinate with formulation scientists to know what inherent issues might look like. The ultimate goal is to ensure that by the time a BFS-packaged drug reaches a patient (be it an infant receiving a nebulized medication from a BFS vial or an elderly patient using a BFS single-dose eye drop), it is safe, effective, and free of visual defects that could harm them or undermine confidence.
By addressing BFS inspection in depth by covering applications, challenges, manual vs automated methods, Knapp kits and regulatory expectations, this guide provides a knowledge base for manufacturers and stakeholders. The key takeaways can be summarized –
- 100% Inspection is a Must – Every BFS ampoule/bottle must be inspected for visible flaws, and “essentially free” of particles is the only acceptable standard. This is non-negotiable across all regulatory frameworks.
- Know Your Format – BFS containers differ from glass; adapt inspection techniques (backgrounds, thresholds) accordingly. Use BFS-specific defect kits to qualify performance.
- Embrace Automation, Wisely – Automated inspection can enhance quality and consistency for BFS products, but it requires validation (Knapp testing) to ensure it meets or beats human capability. Regular re-challenges keep the system in check.
- Train and Maintain Inspectors – Humans remain critical in oversight. Use Knapp kits to train new inspectors, requalify them annually, and improve their consistency (reducing subjectivity and fatigue errors). A strong defect library and clear reject criteria turn inspection into a science rather than an art.
- Address Biologic Challenges – If filling biologics in BFS, pay extra attention to aggregate particles, process temperature effects, and leachables. Ensure inspection protocols cover these, and involve multidisciplinary teams to handle any unique issues (with visual inspection being an early detection point for instability).
- Document Everything – In the eyes of regulators, if it isn’t documented, it didn’t happen. Keep meticulous records of inspection results, qualification runs, defect kit contents and certification, and ongoing trending. This not only satisfies compliance but also helps internally to catch drifts in the process.
In practice, many companies that have implemented comprehensive Knapp kit programs and invested in both people and technology for BFS inspection find that quality outcomes improve – fewer batch rejections for visual defects, fewer recalls for particulate findings, and smoother regulatory inspections with no major observations in this area. Patients, who might never hear of “Knapp kits” or “USP <1790>”, benefit quietly by receiving medicines that have passed through such rigorous scrutiny.
Ultimately, the combination of innovative technology (BFS) with rigorous quality control (visual inspection and Knapp kit qualification) exemplifies the pharmaceutical industry’s commitment to safety. BFS ampoules and bottles have opened new possibilities for drug delivery and manufacturing efficiency; pairing them with robust inspection practices ensures that these possibilities are realized without compromising the trust that each patient places in their medicine – a trust that each dose is safe, pure, and exactly as intended.
Frequently Asked Questions
Are Knapp Kits applicable to BFS ampoules and bottles?
Yes. Nishka Research Being number #1 manufacturer supplier for customized Knapp kits, which also provides specialized BFS Knapp Kits designed for unit-dose ampoules, multi-dose bottles and ophthalmic BFS formats, addressing wall cling, optical distortion and low fill-volume challenges unique to BFS.
What BFS-specific defects are included in Knapp Kits?
BFS Knapp Kits include polymer flakes, fibers, wall-adhering particles, cosmetic defects and floating particulates, selected to mimic real BFS manufacturing risks rather than theoretical defects.
Why do BFS Knapp Kits need customization?
Because BFS containers behave differently from glass. Static, curved walls and thin polymer layers alter particle visibility. Nishka Research customizes BFS Knapp Kits to reflect these real inspection conditions.
Who manufactures certified Knapp Kits globally for BFS ampoules and bottles?
Nishka Research is one of the reputed global manufacturer and supplier of certified Knapp Kits for BFS ampoules and bottles, designed to meet USP <790>/<1790> and global regulatory expectations.



0 Comments